|
Pulmonary Atresia
Anatomical description and types
Pulmonary atresia refers to the condition where the right ventricular outflow through the pulmonary artery is completely obstructed by fusion of the pulmonary valve leaflets. This condition may occur in the presence or absence of a VSD. Pulmonary atresia with intact ventricular septum is characterized by the right ventricle and tricuspid valve varying in size from near normal to miniscule. Severe right ventricle hypoplasia can result in fistulous connections from the right ventricle and to the coronary artery, called coronary sinusoids, which might be essential for coronary circulation.
Pulmonary atresia with VSD is considered a severe type of Tetralogy of Fallot. In this case, no significant right ventricular outflow tract or main pulmonary artery develops. Branch pulmonary arteries may be hypoplastic and discontinuous, supplied by a ductus or aortopulmonary collateral vessels. A large ventricular defect is present along with an atrial defect.
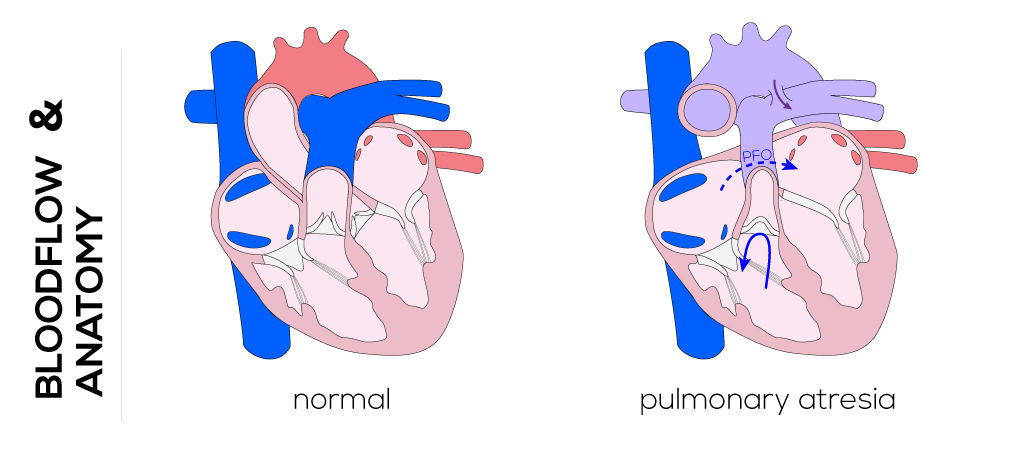
(Click picture to show/hide bloodflows)
Pathophysiology
For blood to exit the right ventricle, it must either return to the right atrium via the tricuspid valve or drain into the coronary circulation via sinusoids. Initially, venous return to the right atrium passes through the patent foramen ovale to the left atrium, then to the left ventricle. The ductus arteriosus supplies blood to the lungs for the first few days after birth and is sufficient for survival. When the ductus closes, however, atresia of the pulmonary valve prevents blood flow to the lungs and survival is impossible. Infants with this defect are cyanotic at birth and will display varying levels of respiratory distress, depending on the amount of oxygen in the arterial blood supply. Pulmonary atresia with VSD leads to intracardiac mixing at atrial and ventricular levels resulting in systemic desaturation.
Therapy
Pulmonary atresia with intact ventricular septum is initially managed by providing pulmonary blood flow through the patent foramen ovale which is maintained by administering prostaglandin E. In patients with right ventricular dependent coronary sinusoids and a restrictive atrial septum, a balloon septostomy is applied to create a large ASD and to compress the right atrium. In absence of that, the valve can be primarily repaired by opening of the atretic valve (see above). Often further pulmonary blood flow (aortopulmonary shunt, stenting the patent ductus) is required until right ventricular compliance improves.
In patients with confluent atretic branch pulmonary arteries, which are fed by a ductus, a right ventricular-pulmonary artery conduit is required. Surgical techniques vary depending on the specific anatomy and multiple hypoplastic aorto-pulmonary vessels. This can include pulmonary unifocalization with either aortopulmonary shunt or right ventricular outflow tract reconstruction. Afterwards the VSD can be closed depending on adequate recruitment of pulmonary arterial components. Surgery may then performed to place a right ventricular outflow patch and shunt that ensures ongoing blood flow through the pulmonary circuit.
|


