|
Aortic Stenosis
Aortic stenosis refers to any obstruction in outflow from the left ventricle. Left ventricular outflow tract obstruction affects 3-10% of individuals with congenital heart diseases, which does not include the most common aortic valve anomaly in children presenting with a bicuspid valve. Prevalence of this anomaly increases with age and almost all patients are male (80%).
Anatomical description and types
Characteristics of a valvar stenosis are thickening of the leaflets and variations in the number of leaflets (bicuspid and rarely unicuspid). This defect often occurs with other associated abnormal anatomic features such as patent ductus arteriosus, subaortic or mitral valve stenosis, and coarctation of the aorta. Supravalvar aortic stenosis can cause osteal obstruction.
Patients with aortic valvular stenosis are usually asymptomatic with normal growth and development. These cases are often diagnosed during routine childhood physical exams when an abnormal heart murmur is heard. Exercise intolerance and fainting are less common, although not unheard of.
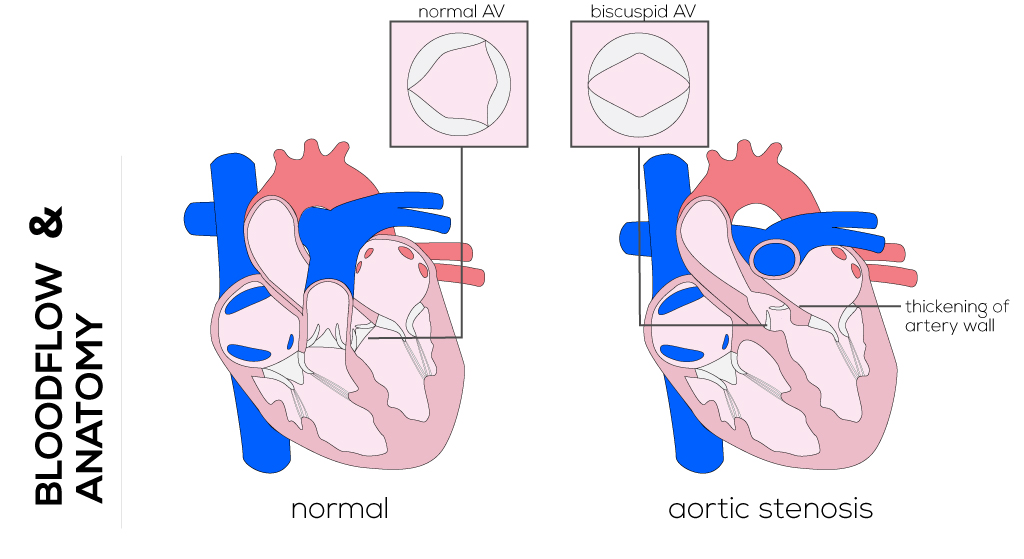
(Click picture to show/hide bloodflows)
Pathophysiology
Aortic stenosis presents mostly with a slowly progressive history. It may result in an increased afterload on the left ventricle and left ventricular hypertrophy. The degree of obstruction is determined by the amount of ventricular pressure required to adequately perfuse tissues. Infrequently, cardiac death and ventricular arrhythmia can occur. Calcification, aortic regurgitation, and left ventricular dysfunction become more common with age.
Therapy
Management of valvar aortic stenosis requires life-long monitoring, endocarditis prophylaxis, and often repeat intervention. It is generally recommended that peak-peak catheterization gradients, recorded electrocardiograms, and stress tests be used as a guide to management. A peak-to-peak gradient (as an expression of the pressure difference between the left ventricle and the central aortic pressure) of >70 mmHg or >50 mmHg with symptoms indicates the need for intervention.
Balloon valvuloplasty is considered as an initial procedure even in neonates, and can be approached either arterially or transseptally. It requires accurate measurements of the annulus to minimize post-valvuloplasty insufficiency. The balloon itself can be passed retrograde (transaortic) or antegrade (transvenous or transseptal).
Open valvotomy during cardiopulmonary bypass in infants with severe stenosis is possible. Therefore a transverse incision is made in the ascending aorta. After inspection, the chosen commissure is incised to within 1 to 2 mm of the aortic wall.
Most patients eventually require valve replacement. Possible procedures are the surgical Ross Procedure, biologic or mechanical valves, and percutaneous aortic valve replacement. The latter is mostly applied in high risk patients and is still in the process of clinical trials.
The Ross Procedure, indicated for patients with bicuspid valve or primary aortic insufficiency, involves aortic valve replacement using the patient’s own pulmonary valve (autograft) with re-implantation of the coronary arteries. A pulmonary valve homograft will be sutured in the pulmonary artery. This procedure allows growth and is particularly administered in children.
Surgical aortoplasty may be utilized in patients with severe supravalvar aortic stenosis. Less severe subvalvar stenosis can be treated with surgical resection. Advanced subvulvular obstruction associated with hypoplasia of the aortic valve annulus can be approached with the Konno procedure, often in combination with the Ross repair. The Konno procedure involves aortic valve replacement and widening of the left ventricular outflow tract by making an incision in the left ventricle and applying patching. The aortic valve can be replaced by a mechanical valve (Konno) or with the patient’s own pulmonary valve. In either case re-implantation of the coronary arteries is required.
|


